 
When, however, one or more domains involve nonbonding pairs of electrons, we must remember to ignore those domains when talking about molecular shape. When all the electron domains in a molecule arise from bonds, the molecular geometry is identical to the electron-domain geometry. From knowing how many of the domains are due to nonbonding pairs, we can then predict the molecular geometry. In determining the shape of any molecule, we first use the VSEPR model to predict the electron-domain geometry. In contrast, the molecular geometry is the arrangement of only the atoms in a molecule or ion-any nonbonding pairs in the molecule are not part of the description of the molecular geometry. The arrangement of electron domains about the central atom of an AB n molecule or ion is called its electron-domain geometry. The shapes of different AB n molecules or ions depend on the number of electron domains surrounding the central atom. If you compare the geometries in Table 9.1 with those in Figure 9.3, you will see that they are the same. These arrangements, together with those for five- and six-electron domains, are summarized in TABLE 9.1. Like the balloons in Figure 9.5, two electron domains orient linearly, three domains orient in a trigonal-planar fashion, and four orient tetrahedrally. In fact, the analogy between electron domains and balloons is so close that the same preferred geometries are found in both cases. The best arrangement of a given number of electron domains is the one that minimizes the repulsions among them. Like the balloons in Figure 9.5, electron domains try to stay out of one another's way. The VSEPR model is based on the idea that electron domains are negatively charged and therefore repel one another. Electron-Domain Geometries as a Function of Number of Electron Domains.Suppose a particular AB 3 molecule has the resonance structureĭoes this structure follow the octet rule? How many electron domains are there around the A atom? In general, each nonbonding pair, single bond, or multiple bond produces a single electron domain around the central atom in a molecule. Thus, the resonance structure for O 3 has three electron domains around the central oxygen atom (a single bond, a double bond, and a nonbonding pair of electrons): For example, the Lewis structure of NH 3 has four electron domains around the central nitrogen atom (three bonding pairs, represented as usual by short lines, and one nonbonding pair, represented by dots):Įach multiple bond in a molecule also constitutes a single electron domain. Likewise, a nonbonding pair (or lone pair) of electrons defines an electron domain that is located principally on one atom. We will refer to such a region as an electron domain. 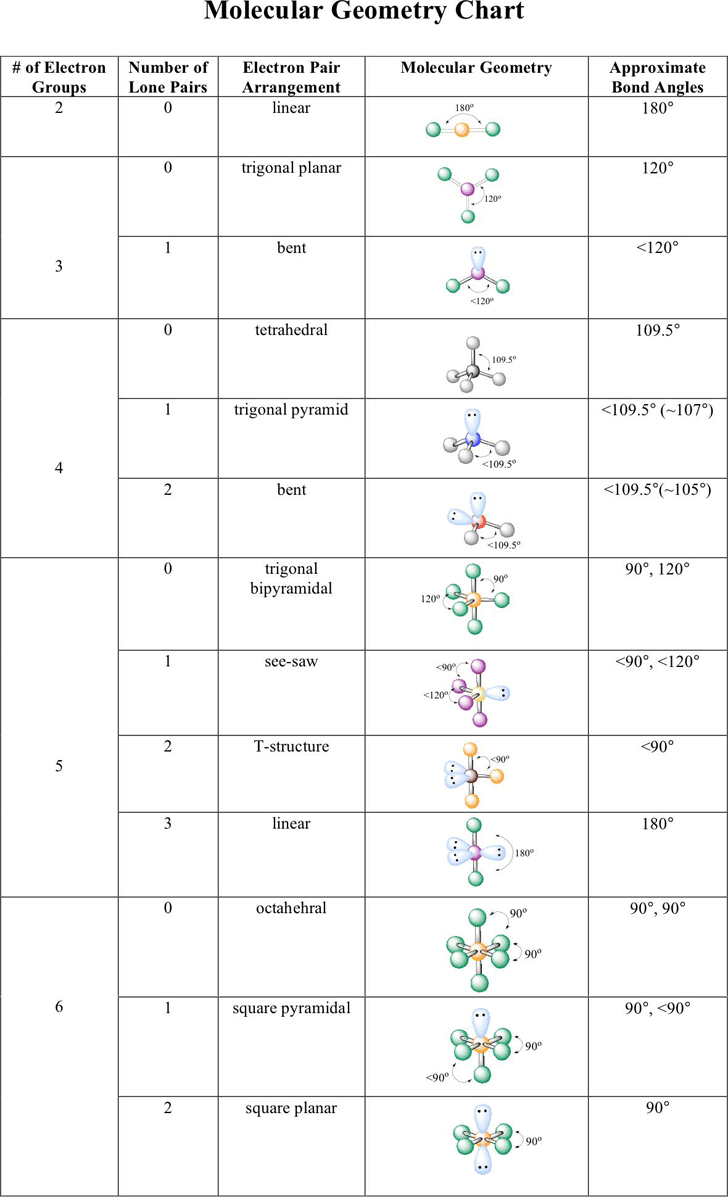
(Section 8.3) A bonding pair of electrons thus defines a region in which the electrons are most likely to be found. We have seen that a single covalent bond is formed between two atoms when a pair of electrons occupies the space between the atoms. In some ways, the electrons in molecules behave like these balloons. We see that an optimum geometry exists for each number of balloons. If we add a third balloon, the balloons orient themselves toward the vertices of an equilateral triangle, and if we add a fourth balloon, they adopt a tetrahedral shape. As shown in FIGURE 9.5, the two balloons naturally orient themselves to point away from each other that is, they try to “get out of each other's way” as much as possible. Imagine tying two identical balloons together at their ends. CHEMISTRY THE CENTRAL SCIENCE 9 MOLECULAR GEOMETRY AND BONDING THEORIES 9.2 THE VSEPR MODEL 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
